Introduction
Cardiovascular diseases (CVDs) are a group of disorders of the heart and blood vessels, and the number one cause of death globally.1Global Burden of Disease study reported in Ethiopia that the mortality in CVD is 18.3% of all deaths in 2016.2Mainly CVDs are caused by chronic kidney disease (CKD).3–5Both CVD and kidney disease are closely interconnected and disease of one organ cause dysfunction of the other,6finally leading to the failure of both organs and this is often referred as a cardiorenal syndrome (CRS).3
CKD is defined as structural or functional abnormalities of the kidney that persist for at least 3 months and is manifested by either kidney damage (most frequently detected as persistent albuminuria or proteinuria (> 30 mg/24h or > 1 on specific dipstick); or a decreased glomerular filtration rate (GFR), (< 60 mL/min per 1.73 m2).7Early CKD has no sign or symptom,8this is why CKD usually remains undetected for a longer period until a screening test identifies the silent problem.
CKD is a global health burden with a high economic cost to health systems9,10and higher among patients with CVD.11,12The factors underlying the increased prevalence of CKD are the progressive aging, diabetes mellitus, and high blood pressure, most of which are present in patients with CVD.4,13–15Blood pressure may be viewed as a continuous variable that fluctuates throughout the day and night in CKD and follows a circadian rhythm with a greater prevalence of daytime hypotension and non/reverse dipper status (ie, higher night-time SBP), which are risky for cardiovascular (CV) events.16,17These fluctuations measured accurately by 24 hr ambulatory blood pressure monitoring, whereas the office blood pressure has some limitations.16,18Hence, blood pressure control is considered a mainstay for slowing the progressive loss of CKD, as well as to lower the risk of CVDs.16,17,19
All stages of CKD are associated with increased risks of CV morbidity, premature mortality, and decreased quality of life. Additionally, CKD is a major and serious risk factor for CVD.7,20Death from CVD is 10 to 30 times higher in dialysis patients than in the general population.21Some studies have confirmed that even early CKD constitutes a significant risk factor for CV events and death.22,23Besides, proper management of CVD is different and more complex in patients with CKD.24
Although the high prevalence of CKD is known in patients with CVD and cause extra morbidity and mortality,22,23,25,26very few studies have quantified CKD and associated risk factors among CV patients in Africa, and no such studies exist in Ethiopia. Also, the findings of the study will help as baseline data for further researches in the future.
Materials And Methods
A hospital-based cross-sectional study was conducted on 163 CV patients at the adult emergency department (ED) of Tikur Anbessa Specialized Hospital (TASH), Addis Ababa, Ethiopia for a period of three months (September to November 2017). The study was conducted following the guidelines laid down in the Declaration of Helsinki and all procedures involving human subjects were approved by the Ethics Committee of College of Health Sciences, Tikur-Anbesa, Medical School, Addis Ababa University. Written informed consent was obtained from all subjects before participation and was informed about the confidentiality of the information they gave. Participants, who were included in this study after being screened by clinicians, were older than 18 years and willing to participate. Exclusion criteria were pregnancy, malnourished, participants less than 18 years old and taking creatine dietary supplements.
Sociodemographic and some risk factor variables were collected using a structured questionnaire. Both bodyweight (kg) and height (cm) without shoes and with light clothes were measured by using a beam balance (Seca, GmbH, Germany). BMI (kg/m2) was calculated from weight in kg divided by height in meters squared (m2). Blood pressure was measured using aneroid sphygmomanometer in the right upper arm in the sitting posture after 30 mins rest. An average of two measurements with five minutes gap between measurements was recorded. Systolic blood pressure (SBP) ≥130 mmHg and/or diastolic blood pressure (DBP) ≥ 80 mmHg or current use of blood pressure-lowering medication was used to define hypertension.27
Five milliliters of the blood sample was collected using a disposable syringe. Serum urea and creatinine were analyzed in the clinical laboratory using an automatic biochemistry analyzer (MINDRAY, BE-2000, CHINA) and urinalysis was done immediately using dipstick dry reagent test strip. The result of 1+ or more was regarded as proteinuria.
An eGFR was calculated separately for men and women using an equation with Modification of Diet in Renal Disease (MDRD):28GFR (expressed in mL/min/1.73 m2) = 186 × [SCr (mg/dl)]–1.154× (age)−0.203× (0.742 if female) × 1.212 (if black). Participants who had eGFR< 60 mL/min/1.73 m2were advised to have their SCr checkup after a month. CKD was categorized according to the classification system guideline of Kidney Disease Outcomes Quality Initiative (K/DOQI). For the purposes of this study, CKD was defined as stages 3–5 CKD (eGFR < 60 mL/min/1.73 m2): with stage 3A (eGFR 45–59.9), 3B (30–44.9), 4 (15–29.9) and 5 (< 15) mL/min/1.73 m2, respectively.29
Data entry and analysis were done using SPSS software version 22. The descriptive statistics were calculated and categorical variables were expressed as frequencies and percentages. Univariate analysis was used to identify risk factors. Multinomial logistic regression was computed for significant variables (p < 0.05) in the univariate analysis. Adjusted odds ratio (AOR) and their corresponding 95% confidence intervals (CI) were expressed to describe the association of independent variables with CKD (dependent variable). Statistical significance was considered at p < 0.05.
Results
Out of 163 CV patients participated in the study, 91 (55.8%) were females. The mean age of the study participants was 42 ± 18 years with 45 ± 18 for males and 40 ± 17 for females. Out of the total patients, 60 (36.8%) were between 25 and 44 years’ age group and 22 (13.5%) were 65 and above years old, ranging from 18 to 86 years old. One hundred ten (67.5%) patients were married and 51 (31.3%) were illiterate. One hundred thirteen (69.3%) patients were urban residents and 74 (45.4%) earned a monthly income of less than 1000 Ethiopian Birr.
Mean BMI of participants was 21.30 ± 4.05 kg/m2and 26 (16.0%) participants were overweight with BMI 25–29.9 kg/m2. Mean SBP and DBP were 105 ± 20 and 67 ± 12 mmHg, respectively, and 52 (31.9%) patients had hypertension. Mean BUN was normal with 18.96 ± 12.54 mg/dl while mean SCr was increased with 1.30 ± 0.96 mg/dl. Mean eGFR values according to MDRD equation was indicated stage 2 CKD with 78 ± 30 mL/min/1.73m2. Among patients diagnosed urinalysis, 41 (25.2%) had proteinuria.
Regarding the distribution of CVDs types, chronic rheumatic valvular heart disease (CRVHD) was the most commonly diagnosed which accounted for 75 (46.0%) followed by Deep Vein Thrombosis 32 (19.6%), Ischemic Heart Disease 19 (11.7%), Stroke 14 (8.6%), Hypertension13 (8.0%), Congestive Heart Disease 3 (1.8%), and others (cardiomyopathies and pericardial disease) 7 (4.3%) in this study.
A total of 124 (76.1%) patients had a normal renal function (eGFR of ≥ 60 mL/min/1.73m2); 23 (14.1%) patients had stage 3a CKD (eGFR 45–59 mL/min/1.73m2); 8 (4.9%) patients had stage 3b CKD (eGFR 30–44 mL/min/1.73m2); and 6 (3.7%) patients had stage 4 or 5 CKD (eGFR <30 mL/min/1.73m2) using MDRD equation. As shown in Table 1, 39 (23.9%) patients had CKD (defined as eGFR < 60 mL/min/1.73 m2).
Table 1 GFR Category According To K/DOQI Classification Using MDRD Equation Among CV Patients Attending At ED In TASH, Addis Ababa, Ethiopia, 2017 |
SBP was significantly associated with CKD (p = 0.023). Although a gender difference in CKD was not significant, it was higher in females compared to males. By age group, older age was not significantly associated with CKD (p = 0.160). CKD was significantly higher among patients with high SCr when compared with low SCr: 21.5% vs 2.5% (p = 0.001). CKD was also significantly higher in patients with high BUN compared with low BUN: 16.6% vs 7.4% (p = 0.001), (Table 2).
Table 2 Distribution Of CKD By Characteristics Of Study Participants Using MDRD Equation Among CV Patients Attending At ED In TASH, Addis Ababa, Ethiopia, 2017 |
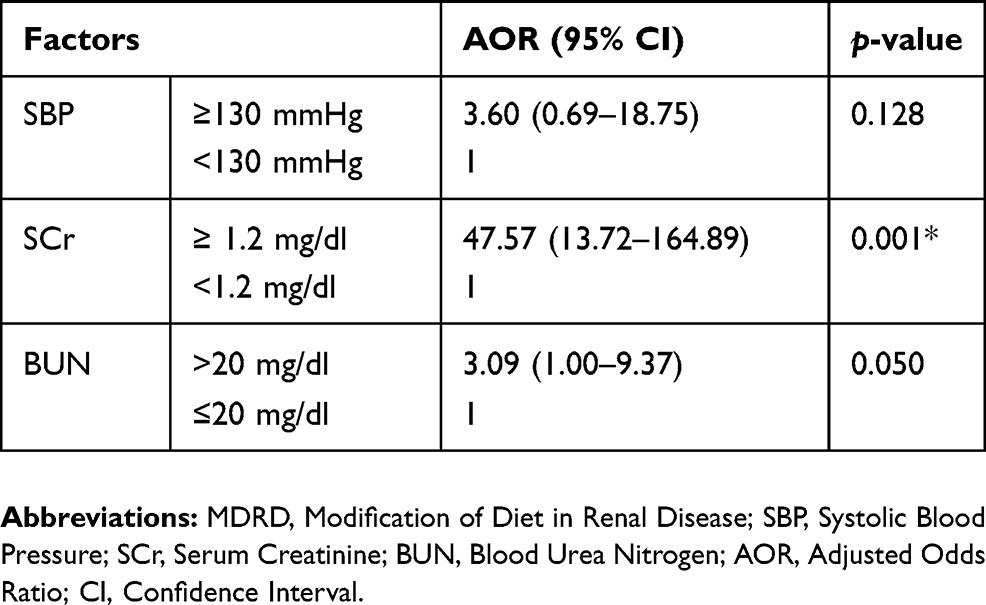
The univariate analysis showed a significant association between CKD (eGFR < 60 mL/min/1.73 m2) and the following variables: elevated SBP, high SCr, and high BUN. After incorporating all significant (p < 0.05) variables in the univariate analysis, multivariate logistic regression was computed to identify risk factors independently associated with CKD. In multivariate analysis, only high SCr (AOR = 47.57, CI 13.72–164.89) was independently associated with CKD (Table 3).
Table 3 Factors Associated With CKD According To The MDRD Equation Among CV Patients Attending At ED In TASH, Addis Ababa, Ethiopia, 2017 |
Discussion
Chronic kidney disease (CKD) is a global health burden with a high economic cost to health systems9,10and higher among patients with CVD.11,12All stages of CKD are associated with increased risks of CV morbidity, premature mortality, and/or decreased quality of life.7,23,26Thus, early detection and recognition of CKD are important in patients with CVD to improve adverse outcomes, delay the progression to end-stage renal disease (ESRD) and encourage early referral to the nephrologist.15
In this study, CKD (eGFR < 60 mL/min/1.73 m2) was found in 23.9% of participants based on the MDRD formula used to estimate GFR, with stage 3 being dominant (19.0%). The estimated prevalence of CKD was lower than reported by Amenos et al15and Yang et al6These differences might be due to the study design and sample size.
The present study showed that an increase in SCr was independently associated with CKD (p= 0.001). The study done by Pandya et al showed that the serum creatinine is highly statistically significant with CKD.30Unlike urea, creatinine is not toxic waste, but simply it is used as a marker of renal function. Although the mechanism by which an increase in SCr cause CKD is unidentified, it used to indicate that 50% of GFR had already been lost.31
In this study, BUN showed a highly significant association with CKD defined by MDRD (p = 0.001). This is consistent with the study of Amsalem et al32Concomitant elevations of BUN implies renal excretory failure, but only at an advanced stage of kidney damage.13BUN is an imperfect measurement of kidney function and is influenced by factors other than GFR.33An elevated BUN can further reflect a state of renal hypoperfusion from hypovolemia, renovascular disease, or reduced cardiac output.34,35BUN may also be raised independently of a change in GFR or SCr due to enhanced urea reabsorption under the activation of the SNS and RAAS.36
Regarding risk factors, this study found an insignificant association between older age and CKD defined by MDRD (p = 0.160). This contradicts with the finding of studies.37,38This might be due to the small sample size used in our study. In fact, younger people have a higher GFR than older people, which may lead to a late diagnosis of kidney disease. As age increases, there is a gradual decrement in the number of nephrons and GFR. Thus, screening CKD in this age group is an important strategy to improve the outcomes.39
In this study, the self-responsive hypertension was not significantly associated with CKD (p = 0.075) although SBP was significantly associated with CKD defined by MDRD equation (p = 0.023). This contradicts with the finding of Chen et al4and Amenos et al15The differences might be due to many patients didn`t know whether they are hypertensive or not and our patients were mainly less than 60 years old. Nowadays, some studies suggest the greater use of antihypertensive medications and specific drug classes such as diuretics, angiotensin converting enzyme inhibitors, and angiotensin II receptor blockers to control blood pressure may affect kidney function independently of their effect on blood pressure.29,40,41In younger people, there is high early vascular damage and particularly larger artery damage in young ages, which increases hypertension.42,43The possible relation is that systemic hypertension is passed on to intraglomerular capillary pressure leading to glomerulosclerosis and loss of kidney function.14Thus, the advantageous effects of controlling blood pressure in CVD has been pronounced repeatedly in current guidelines.27
Although this study is the first time in Ethiopia, it has a few limitations. First, this study was a cross-sectional study, which does not enable those patients with temporary disorders in renal function to be distinguished from those with true CKD. Second, the dipstick provides only a semi-quantitative measurement of proteinuria, relatively insensitive and does not register as positive until total protein excretion is more than 300 mg/day. Third, the influence of other medications and diet were also not taken into consideration during this study. Fourth, we used a routine manual office blood pressure measurement that is on average 9/6 mm Hg higher than standardized blood pressure measurement (24 hr ambulatory blood pressure monitoring).18Our study also has strengths, including assessing renal function by using eGFR and proteinuria in CVD, since it is not studied and explored in Ethiopia.
Conclusion
This finding indicated that the CKD, which was observed in 23.9% of CV patients, was significantly associated with SBP and increased BUN, while independently associated with increased SCr. Thus, early detection and recognition of CKD using eGFR is essential in patients with CVD to avoid extra morbidity and mortality. We recommend using MDRD formula in hearth facilities for diagnosing of CKD to reduce duplication of laboratory test (SCr and BUN), which is the easiest practice and save costs in patient’s pockets and public sectors.
